Human cytomegalovirus (HCMV) has a large (~235-kb) genome with over 170 ORFs and exploits numerous cellular factors to facilitate its replication. HCMV infection increases protein levels of WD repeat-containing protein 5 (WDR5) during infection, overexpression of WDR5 enhances viral replication, and knockdown of WDR5 dramatically attenuates viral replication. WDR5 is essential for assembling the VISA-associated complex to induce a type I interferon antiviral response to Sendai virus infection. However, the roles of WDR5 in DNA virus infections are not well described.
In a present study, the research group led by Prof. LUO Minhua in Wuhan Institute of Virology of the Chinese Academy of Sciences indicated that WDR5 promotes nuclear egress of viral capsids, resulting in significant decrease in production of infectious virions. This is the first report that WDR5 favors HCMV, a DNA virus replication, and highlights a novel target for anti-viral therapy.
In this study, the scientists found that WDR5 expression increases during HCMV infection and this serves to enhance HCMV replication. Furthermore, knockdown of WDR5 resulted in defects in capsid nuclear egress and reduced production of infectious virions. Their findings suggest that WDR5 contributes to HCMV replication.
They found that HCMV infection increased expression and stabilized the cellular protein WDR5. Overexpression of WDR5 slightly enhanced HCMV replication while knockdown significantly attenuated HCMV replication. To identify the steps in which WDR5 is involved, the replicative cycle of HCMV replication was investigated in WDR5-depleted cells. Their data indicate that formation of the NEC and IINMs were significantly impaired, suggesting that nuclear egress of HCMV capsids is a key step in which WDR5 is involved.
In summary, depletion of WDR5 significantly attenuates HCMV replication by decreasing NEC and IINM formation, thereby impairing capsid nuclear egress and, consequently, virion maturation in cytoplasm. These results indicate that WDR5 contributes to HCMV replication via promoting nuclear egress.
The results have been published in Journal of Virology entitled "WDR5 Facilitates Human Cytomegalovirus Replication by Promoting Capsid Nuclear Egress".
This work was supported by grants from the National Natural Science Foundation of China, Key Basic Research Project from MOST, Sino-Africa Joint Research Center, Chinese Academy of Sciences, and the Key Program of Guangzhou Joint Healthcare Innovation Foundation.
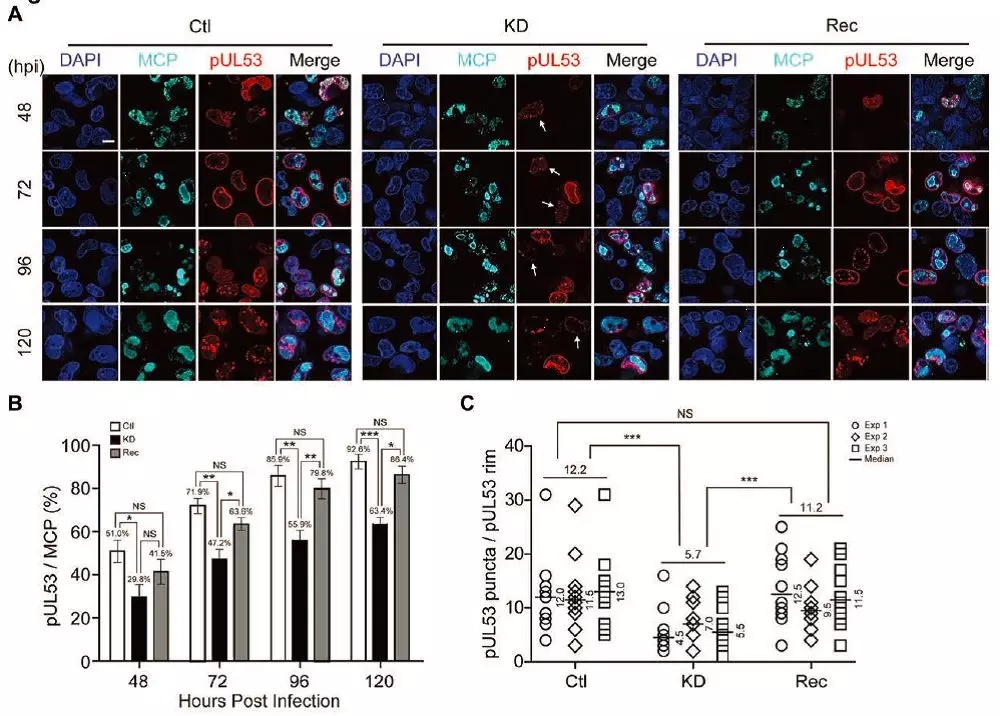
Knockdown of WDR5 affects capsid nuclear egress by impairing NEC formation. Image by LUO Minhua
Contact:
LUO Minhua
E-mail: luomh@wh.iov.cn
Wuhan Institute of Virology, Chinese Academy of Sciences, Wuhan 430071, China (http://english.whiov.cas.cn/)