Scientists revealed that Ac102 and C42 form a regulatory cascade to control viral NPF activity
Date:07-05-2018 | 【Print】 【close】
Actin is one of the most functionally important proteins in eukaryotic cells. Morphologically, actin can be found in two forms: a monomeric form called globular actin (G-actin) and a polymeric form called filamentous actin (F-actin).
G-actin can polymerize to form F-actin, and nucleation promoting factor (NPF) is the initiator of this process. Many viral pathogens harness the host actin polymerization machinery to assist in virus propagation. Autographa californica multiple nucleopolyhedrovirus (AcMNPV) induces actin polymerization in host cells. P78/83, a viral NPF, is responsible for this process. Previously, the scientists identified that BV/ODV-C42 (C42) binds to P78/83 and protects it from degradation.
In a recent report, the research group led by Prof. CHEN Xinwen in Wuhan Institute of Virology of the Chinese Academy of Sciences determined that another viral protein, Ac102, is involved in modulating C42 ubiquitination and consequently ensures P78/83 activity as an NPF to initiate actin polymerization. This regulatory cascade represents a novel mechanism by which a virus can harness the cellular actin cytoskeleton to assist in viral propagation.
In this study, the scientists identified that Ac102 is a novel regulator of actin polymerization during AcMNPV infection. Knockout of ac102 from the AcMNPV genome resulted in nuclear actin polymerization deficiency, as well as abnormal morphogenesis and distribution of capsid structures in the nucleus. These phenotypical changes are heavily dependent on the Ac102-C42 interaction but are not correlated with the nuclear accumulation of actin. Further investigation indicated that Ac102 suppresses K48-linked ubiquitination of C42 and consequently potentiates P78/83 availability as an NPF to induce actin polymerization.
In summary, their study revealed a novel regulatory cascade consisting of Ac102 and C42, which extends our understanding of how baculovirus manipulates the host actin polymerization machinery to maximize its value to viral propagation.
The results have been published in Journal of Virology entitled "Ac102 Participates in Nuclear Actin Polymerization by Modulating BV/ODV-C42 Ubiquitination during Autographa californica Multiple Nucleopolyhedrovirus Infection".
This work was supported by grants from the National Natural Science Foundation of China.
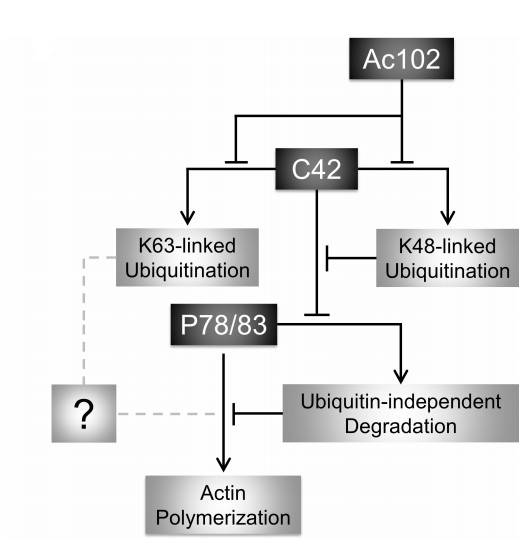
Model of a regulatory cascade that controls actin polymerization during AcMNPV infection. Image by CHEN Xinwen
Contact:
CHEN Xinwen
E-mail: chenxw@wh.iov.cn
Wuhan Institute of Virology, Chinese Academy of Sciences, Wuhan 430071, China (http://english.whiov.cas.cn/)