Researchers present a structure of RNA polymerase complex and genome within a dsRNA virus
Date:04-07-2018 | 【Print】 【close】
Double-stranded RNA (dsRNA) viruses infect hosts ranging from fungi to plants and to humans. Like most other dsRNA viruses, the aquareovirus RNA polymerase catalyzes the synthesis of RNA plus strands within the inner capsid, a process vital for the replication of virus progeny.
In a recent study, by cooperation with Tsinghua University, the research group led by Prof. FANF Qin in Wuhan Institute of Virology of the Chinese Academy of Sciences presented a near-atomic resolution structure of the RNA polymerase in complex with its cofactor protein and genomic RNA within the aquareovirus. Some asymmetric structures within the elaborate viral machine that have never been previously determined have been resolved in their structure, and key interactions among the polymerase, cofactor protein, and dsRNA have been revealed.
In this study, the scientists determined the structure of the RdRp protein VP2 in complex with its cofactor protein VP4 and genomic RNA within the double-layered aquareovirus capsid using cryoelectron microscopy (cryo-EM) at 200 kV and our symmetry-mismatch reconstruction method.
Their structure shows that the VP2–VP4 complex is anchored at the capsid shell and interacts with genomic dsRNA and four of the five asymmetrically arranged N termini of capsid shell protein VP3A under the fivefold axis, implying roles for these N termini in virus assembly. In addition, some elements in the VP2, which is supposed to interact with template RNA and priming nucleoside triphosphate (NTP) during transcription, are flexible.
A loop, which is thought to separate RNA template and transcript, was observed to interact with an apical domain of the shell protein VP3A, suggesting that the conformational change of the apical domain upon virus transcription activation could regulate RdRp replication and transcription. A conserved NTP binding site was localized in the VP4 structure, and both VP4 N-terminal and C- terminal domains are interacting with the genomic RNA.
These findings provide insights into the mechanism underlying highly coordinated dsRNA virus transcription and assembly. The results have been published in PNAS entitled "Structure of RNA polymerase complex and genome within a dsRNA virus provides insights into the mechanisms of transcription and assembly".
The study was supported by the National Research and Development Program of China, the National Natural Science Foundation of China, and Natural Science Foundation of Hunan Province.
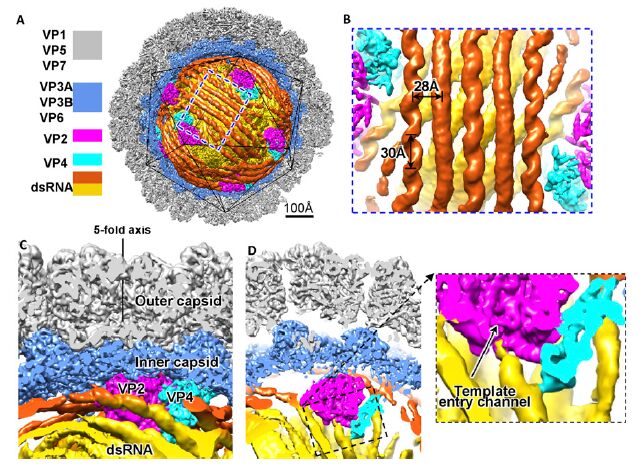
Structures of the aquareovirus genome and RdRp complex within the capsid. Image by FANG Qin.
Contact:
FANG Qin
E-mail: qfang@wh.iov.cn
Wuhan Institute of Virology, Chinese Academy of Sciences, Wuhan 430071, China (http)://english.whiov.cas.cn/