Scientists provide the first demonstration of the NTPase/helicase-like activity encoded by EBOV
Date:04-06-2019 | 【Print】 【close】
Ebola virus (EBOV) is a filamentous, enveloped, non-segmented, negative-sense RNA virus, belonging to the genus Ebolavirus in the family Filoviridae of the order Mononegavirales. From 2014 to 2016, West Africa has experienced the largest Ebola outbreak in the history that has resulted in more than 28000 cases and over 11000 deaths.
Moreover, the most recent Ebola outbreak is still on-going and deteriorating in DR Congo, which has caused 1290 EVD cases and 833 deaths until 16 April 2019. Thus far, no approved drug or vaccine is commercially available against EBOV.
In a recent study, the research group led by Prof. ZHOU Xi in Wuhan Institute of Virology of the Chinese Academy of Sciences reported for the first time that the EBOV-encoded multifunctional protein VP35 has NTPase and helicase-like activities that can bind to NTPs, hydrolyze all types of NTPs and unwind RNA helices in an NTP-dependent manner.
Moreover, their work showed that GuHCl, an FDA-approved small molecule drug, is able to inhibit the NTPase/helicase-like activities of VP35 in vitro, and more importantly, the replication and transcription of a stable EBOV minigenome replicon in cultured human cells.
In conclusion, this study provides the first demonstration of the NTPase/helicase-like activity associated with an NNSV-encoded protein, EBOV VP35, and finds that GuHCl is an inhibitor of not only the in vitro NTPase and helicase-like activities of EBOV VP35, but also the replication/transcription of EBOV minigenome in human cells.
These findings uncover an unexpected novel function of EBOV VP35, extend the view of RNA remodelling proteins, provide the evidence that guanidine-containing compounds may serve as potential candidates for anti-EBOV/filovirus drug development by targeting VP35, and shed light on the understanding of EBOV and filovirus replication and pathogenesis.
Their study was published in Nucleic Acid Research entitled “Ebola virus VP35 has novel NTPase and helicase-like activities”.
This work was supported by Strategic Priority Research Program of Chinese Academy of Sciences, National Science and Technology Major Project, National Natural Science Foundation of China, Newton Advanced Fellowship from the Academy of Medical Sciences, UK, Science and Technology Bureau of Wuhan, and Advanced Customer Cultivation Project of Wuhan National Biosafety Laboratory.
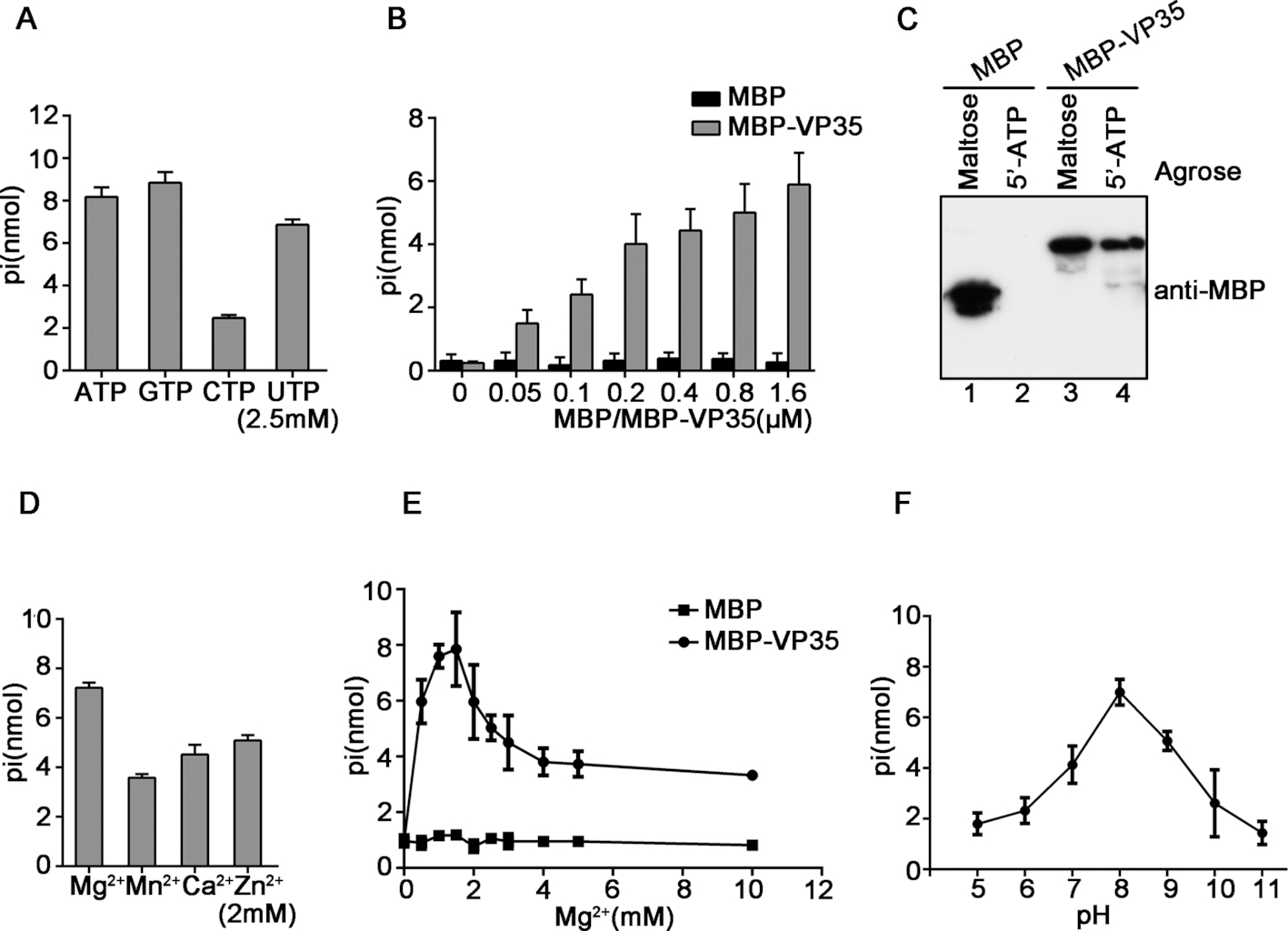
EBOV VP35 has NTP-binding and NTPase activities. Image by ZHOU Xi
Contact:
ZHOU Xi
E-mail: zhouxi@wh.iov.cn
Wuhan Institute of Virology, Chinese Academy of Sciences, Wuhan 430071, China. (http://english.whiov.cas.cn/)